|
8/17/2023 0 Comments Pathological hypertrophy heart  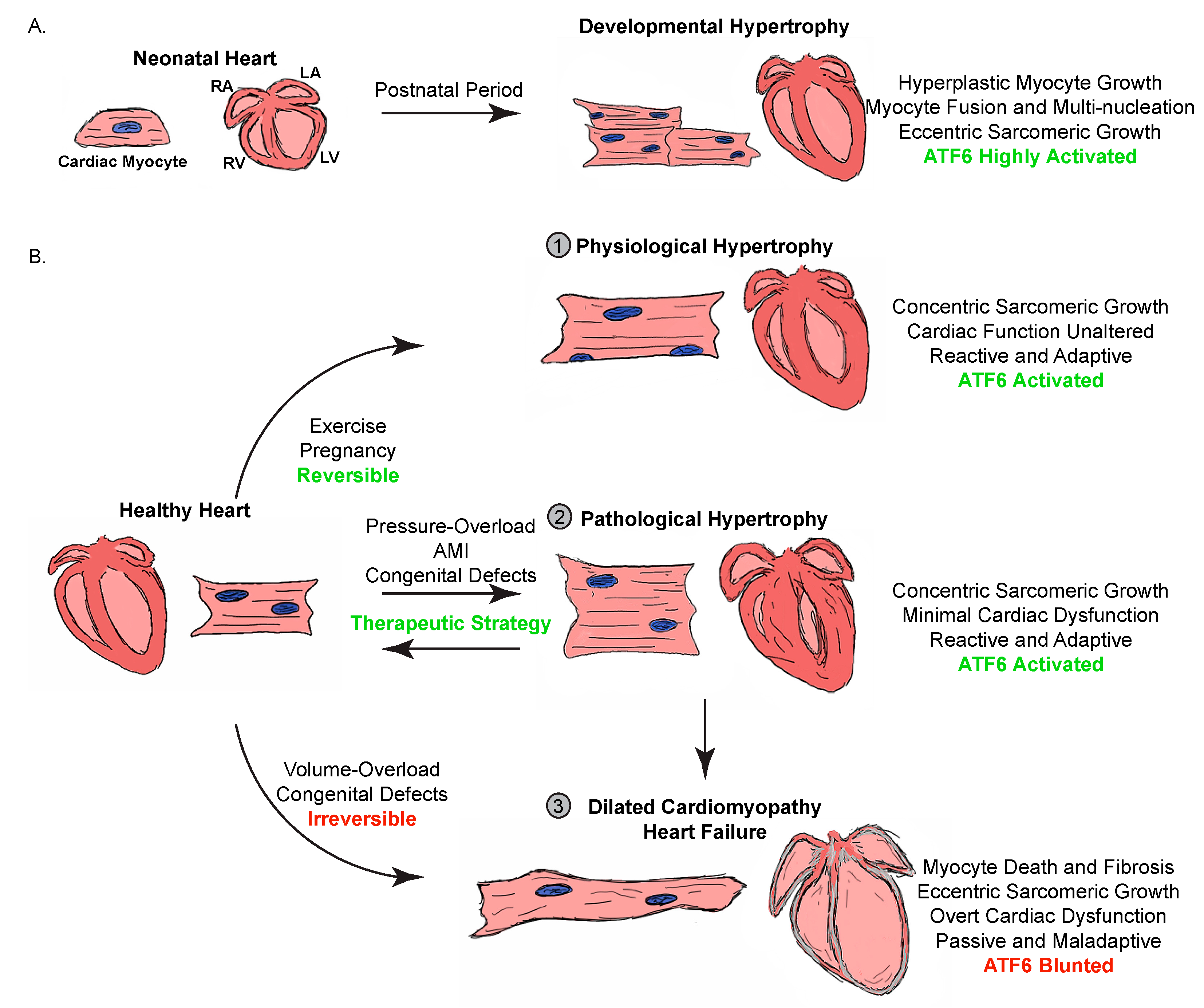
Heart targeted knockout studies have implicated BRAF in pathological hypertrophy and interstitial fibrosis. Contractility denotes contractility and/or cardiac function. The symbol ‘*’ indicates that only gene expression was reported. Red text indicates pathological hypertrophy. None of the gain-in-function studies for BRAF enhanced cardiac fibrosis, although loss of BRAF attenuated fibrosis under pathological conditions. Inexplicably, pharmacological activation of BRAF attenuated Ang II-induced pathological hypertrophy (as assessed by gene expression) and improved cardiac contractility/function. Pharmacological activation of BRAF (the RAF paradox via Type 1 RAF inhibitors) had similar effects with the exception of pathological gene expression. Expression of a targeted knockin activated BRAF mutant was found to induce hypertrophy associated with an increase in markers of pathological hypertrophy, although contractility/cardiac function improved. Divergent results were reported for the loss of BRAF on physiological hypertrophy in male versus female mice, but these results are complicated by a reduction in RAF1 in male mice only. These studies were performed on male mice only and a sex difference was not studied. Summary of studies implicating BRAF in cardiac hypertrophy and fibrosis Nevertheless, the new findings highlight the potential importance of additional signaling events, perhaps related to RAF1 or ERK1/2 activation, in shaping BRAF signaling in a sex- and context-dependent manner. In addition, the role of BRAF and its activation in overt heart failure remains to be established. Additional studies are needed to better define the role of BRAF under conditions reflective of chronic stress on the heart due to the biomechanical stimulation exerted by hypertension. These observations extend those previously made by this group assessing the consequences of expressing an inducible activating mutant of BRAF in the heart and the benefit of enhancing RAF/MEK/ERK1/2 signaling by exploiting the ‘RAF paradox’. In contrast, loss of BRAF attenuated physiological hypertrophy in female mice but had no effect on fibrosis or contractility. BRAF knockout decreased cardiac function with PE in male mice and enhanced both interstitial and perivascular cardiac fibrosis but had no effect on hypertrophy. They reported that loss of BRAF attenuated both pathological cardiac hypertrophy and interstitial fibrosis. utilized an inducible cardiac myocyte targeted knockout mouse model to define the role of BRAF in pathological versus physiological hypertrophy using angiotensin II and phenylephrine (PE) infusion, respectively. In a recent issue of Clinical Science, Alharbi et al. The RAF family of kinases consists of three isoforms (ARAF, BRAF, and CRAF) and until recently most studies on this signaling pathway in the heart have focused on RAF1 (CRAF). All rights reserved.The RAF/MEK/ERK1/2 signaling cascade has been implicated in pathological cardiac hypertrophy downstream of some Gq-coupled receptors. This review describes the key molecules and cellular responses involved in physiological/pathological cardiac hypertrophy.Īkt Angiogenesis Autophagy Cardiac hypertrophy Epigenetic modification Heart failure Inflammation Metabolism MicroRNA.Ĭopyright © 2016 Elsevier Ltd. Pathological hypertrophy is associated with fibrosis, capillary rarefaction, increased production of pro-inflammatory cytokines, and cellular dysfunction (impairment of signaling, suppression of autophagy, and abnormal cardiomyocyte/non-cardiomyocyte interactions), as well as undesirable epigenetic changes, with these complex responses leading to maladaptive cardiac remodeling and heart failure. In contrast, pathological hypertrophy is induced by factors such as prolonged and abnormal hemodynamic stress, due to hypertension, myocardial infarction etc. Physiological hypertrophy of the heart occurs in response to normal growth of children or during pregnancy, as well as in athletes. Cardiac hypertrophy is classified as physiological when it is associated with normal cardiac function or as pathological when associated with cardiac dysfunction. The processes of growth (hypertrophy), angiogenesis, and metabolic plasticity are critically involved in maintenance of cardiac homeostasis. To maintain the high energy consumption required by this role, the heart is equipped with multiple complex biological systems that allow adaptation to changes of systemic demand. 
The heart must continuously pump blood to supply the body with oxygen and nutrients. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
